Corrosion resistance in zinc die casting is not dependent on the alloy but on how well the casting is able to be protected by the environment in which it operates through surface treatment.
It is still common practice among many OEM engineers and sourcing managers to expect zinc alloys to be intrinsically zinc oxide-based corrosion-proof metallic components. Practically untreated or unfinished zinc die castings are usually white superficially corroded, pitted, or corroded in months in damp, saline or chemically hostile environments. In actual life scenarios, such as the automotive brackets that are exposed to road salt, or the electronics housing that are exposed in coastal regions, the actual service life will depend on the engineered protection of the surface that is used as a defensive barrier, a sacrificial one, or both.
Even high purity zinc alloys (with extremely high impurity levels such as those of the Zamak or ZA series) are not evenly corroded, without properly treating the surface, where moisture, chlorides, or pH extremes enter microscopic porosities or local defects of many die castings. That is why the integrated process control which is achieved by designing molds up to finishing is so important. In case, our experience with precision zinc die casting parts manufacturing shows that combining tight process controls with the right post-casting treatment consistently delivers parts that withstand years of demanding exposure without functional loss.
Why Corrosion Resistance Matters for Zinc Die Casting Parts
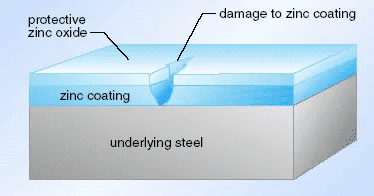
Corrosion Does not limit itself to cosmetic problems with zinc die cast – it directly affects the integrity of parts, their operation and overall cost of ownership.
As the corrosion progresses, it also targets the load-bearing parts of the company, builds stress risers, or results in changes of size or shape that can lead to decreased dimension of assembly fit. Even small pitting may result in early failure of a functional application such as automotive housing or medical devices, warranty or even safety concerns. On an economic basis, corrosion-related rework, scrapping, or repair in the field could cost much more, with downtime and liability included, that first-cost multiplied 5-10 times.
A sketchy list of the common effects is below:
| Corrosion Impact | Functional Risk | Business Consequence |
| Surface pitting / white rust | Aesthetic degradation, reduced conductivity | Customer complaints, brand damage |
| Intergranular attack (rare but severe if impurities present) | Loss of ductility and strength | Catastrophic failure in structural parts |
| Coating delamination | Accelerated base metal corrosion | Increased warranty returns, field failures |
| Thickness loss over time | Dimensional drift, poor fit in assemblies | Assembly line rejects, higher scrap rates |
The results of such negligence by engineers can be reduced service life and increased lifecycle cost, something that we have observed on numerous occasions can be overcome by, in other words, laying emphasis on corrosion strategy early in the design process.
Natural Corrosion Behavior of Zinc Alloys
In most environmental scenarios, zinc alloys develop a stable and adhering layer of oxide/carbonate that retards further corrosion relative to most of the ferrous metals.
This coating -a passive coating, consisting primarily of zinc oxide and basic zinc carbonate, and in light environments of the indoors or countryside, sometimes performs better than bare steel without finishes. When exposed to neutral pH conditions (that is, without exposure to chloride) and without significant graying, untreated Zamak-type castings have a service life of decades.
But there are definite boundaries of natural protection. The humidity hastens the development of white rust (hydrated zinc corrosion products), whereas the salt spray or industrial pollutants (sulfur compounds, chlorides) destroy the layer quickly, which results in pitting. The oxide is preferentially dissolved by acidic (pH <6) or strongly alkaline (pH >12) conditions and any axial attack is made worse by trapped moisture in porosities. Cold castings of zinc seldom last longer than 1212 months in either sea or highway salt conditions, falling very short of estimated OEM zinc life projections.
How Surface Treatments Improve Corrosion Resistance
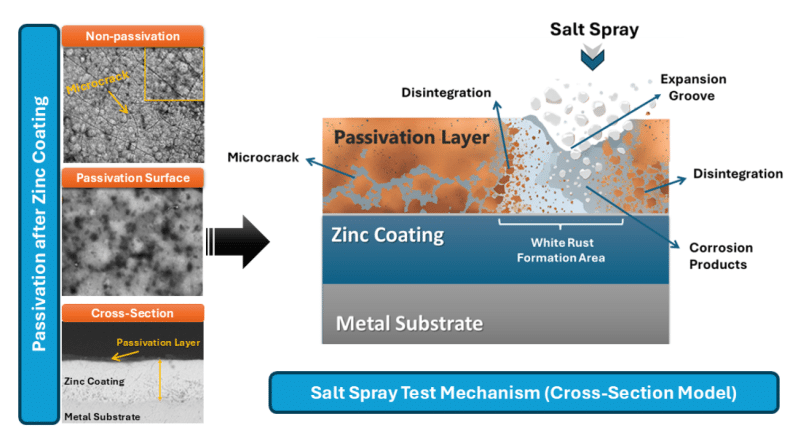
Right surface treatment has been known to convert moderately protected Zinc die casting to highly resistant by applying efficient protection barrier or by using a combination of barrier as well as sacrificial mechanisms.
Physical isolation of the substrate can be done by the use of a barrier coating (e.g. paints, powder coatings), which can serve as a physical isolate to put the substrate out of contact with oxygen, moisture and corrosives or sacrificial barrier (e.g., zinc-rich platings) which will corrode preferentially to protect any bare areas. The continuity of the adhesion as well as coating is important – any interruption(s) (poor preparation or thinness) acts as one point of coresion inception.
Frequent interventions and their results:
| Surface Treatment Type | Protection Mechanism | Typical Corrosion Performance |
| Chromate conversion (clear/iridescent) | Conversion layer + passivation | Good in mild atmospheres; 200–500 h salt spray |
| Electroplating (Cu/Ni/Cr) | Multi-layer barrier (nickel primary) | Excellent; 500–1000+ h salt spray (thickness-dependent) |
| Powder coating / painting | Thick organic barrier | Very good in industrial/marine; 500–1500 h |
| Zinc plating + chromate | Sacrificial + passivation | Moderate to good; self-healing in scratches |
| Anodizing (specialized) | Enhanced oxide thickness | Superior in harsh conditions; marine-grade |
Multi-layer electroplating can frequently prove to be the best balance in our experience of production in that decorative + functional parts are required, and powder coating is required on large volumes, and cost-sensitive, of required parts requires a very strong barrier protection.
Matching Surface Treatment to Application Environment
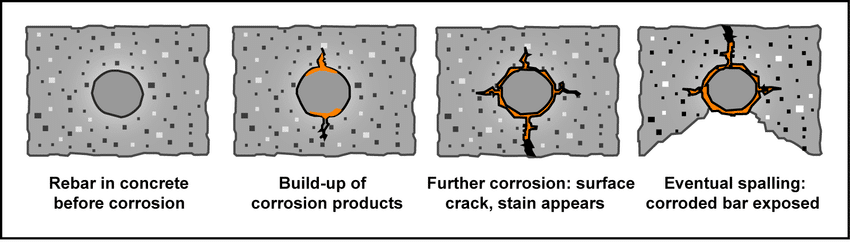
However, end-use environment needs differ markedly in terms of corrosion: an engineer needs to select both corrosion treatment to fit the degree of exposure, not to fall back to a single, so-called universal finish.
The automotive parts are subjected to road salt, temperature fluctuations, and oils and are required to have high salt-sprays performances. Housings of consumer electronics address humidity indoors and spillage infrequently. Components of lighting and appliances tend to focus on aesthetics and moderate exterior or interior while being outdoors or in a kitchen.
For tailored guidance:
- Automotive zinc die casting parts → Require robust plating or powder coating for salt resistance.
- Zinc die casting for consumer products → Often chromate + clear coat for indoor reliability.
- Zinc die casting parts for lighting and home appliances → Balance decorative chrome with underlying corrosion barriers.
Decision framework:
| Application Environment | Corrosion Risk Level | Recommended Surface Treatment |
| Indoor / dry (electronics) | Low | Clear chromate or light electroplate |
| Humid / occasional moisture | Medium | Powder coating or zinc plating + chromate |
| Outdoor / automotive (salt) | High | Multi-layer Ni/Cr plating or thick powder coat |
| Marine / chemical exposure | Very High | Specialized anodizing or heavy barrier systems |
Common Surface Treatment Failures and Their Root Causes
Majority of the failures of coating zinc die castings are caused by failure in the preparation or application and not in the treatment itself.
Incompleteness of surface preparation (poor cleaning of surface, presence of oils or oxides) results in absence of adhesion and consequent blistering or delamination with relation to humidity or thermal cycles. Poor thickness exposes thin areas to the penetration. The design characteristics, such as obscure holes or depressions, entrap pretreatment chemicals or moisture, promoting corrosion at the localities.
In reality, we have found 7080% of complaints in the fields to be linked to prep shortcut procedures: insisting on comprehensive alkaline cleaning; acid etching; and thorough rinsing followed by any coating procedure.
Design Considerations That Influence Corrosion Protection
Effective corrosion behavior begins with the design process- very minor geometry changes may radically enhance the effectiveness of the coating.
Make sure that there are sufficient draft angles and radii to prevent the thinning away at edges. Include drainage aisles to eliminate water stagnancy. Shallows A deep recess will entrap pretreatment solutions or form galvanic couples utilizing fasteners.
| Design Feature | Corrosion Risk | Preventive Design Approach |
| Sharp edges / thin walls | Coating thinning → early breakthrough | Add generous radii (min. 0.5 mm) |
| Blind holes / recesses | Trapped moisture / chemicals | Add vent/escape features or through-holes |
| Dissimilar metal interfaces | Galvanic acceleration | Isolate with seals or compatible fasteners |
| Poor drainage | Standing water → accelerated attack | Slope surfaces, add weep holes |
Common OEM Misconceptions About Corrosion Resistance
There are a number of recurring myths that result in unguarded sections and premature failure.
First there is the fallacy of collecting similar protection in all surface treatments, which just plain overlooks colossal differences chromate by itself is not going to compete with multi-layered plating in salt spray. Second, the assumption of the benefit of greater corrosion resistance that thinner coatings imply ignores the adhesion and consistency; too thick layers might develop cracks. Lastly, those that consider corrosion protection as an almost purely finishing matter are missing the interplay between casting quality, purity of alloys, and design.
Conclusion — Corrosion Resistance Is an Engineered Outcome
Zinc die casting resistance to corrosion is a result of system level choices: selection of alloy, process control, preparation of surface, coating selection, and environment design. OEMs realize predictable high duration of the early assessment of exposure conditions and treating them upfront will provide long, predictable internally consistent durability without excessive over-engineering. You have to treat it as an engineering requirement rather than after-thought and the beneficiaries are P.O.W.
